 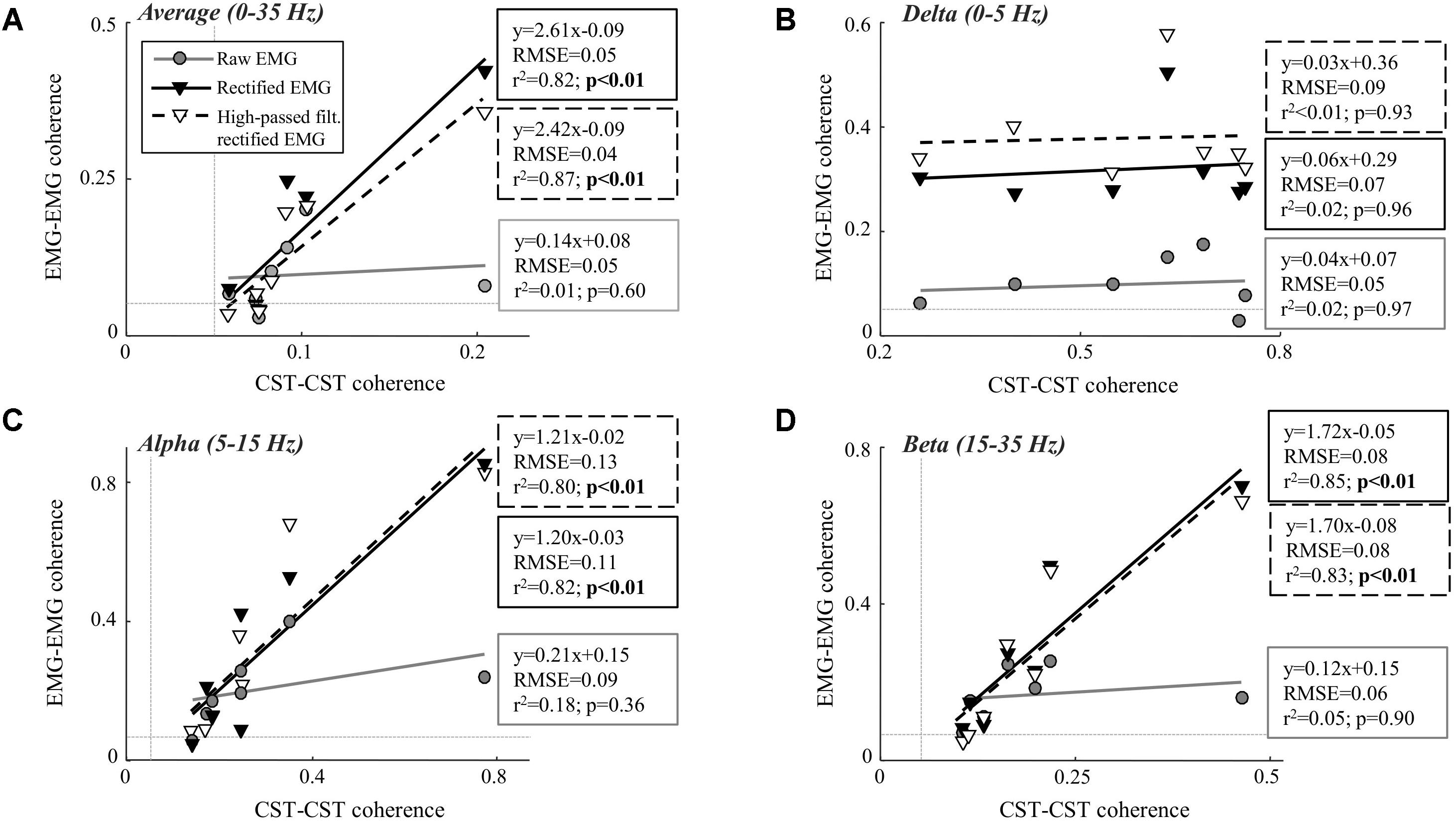 
We investigate this mechanism for memory storage and retrieval but it could also pertain to rapid attention switching 17, 18, 19. Thus a given oscillatory band acts as a readout carrier for neurons that are selectively depolarized and shifts their resonance frequency into that band. We hypothesize that sub-threshold depolarization from synaptic coupling or external input can shift neurons into and out of resonance with specific bands of extracellular oscillations and this resonance shift can act as a mechanism to selectively activate functionally diverse neural populations. We propose a novel mechanism that links the voltage-dependent resonance frequency shifts of individual neurons with the large scale oscillatory rhythms observed in the brain to selectively activate neuronal ensembles. This idea is experimentally supported by findings that oscillations in the visual system can affect downstream processing, as seen in the phase locking of neurons in the LGN to the 50 Hz oscillations in the input received from the retina 16. Sub-threshold oscillatory input through synaptic or ephaptic coupling provides feedback between brain oscillations in the local field potential and individual neurons 15. However, the influence of these oscillations on individual neuronal activity patterns or network wide activity has remained unclear 14. These brain rhythms have been implicated in various cognitive functions 12, 13. The observed resonance shifts range from a couple of Hz to more then 10 Hz.Īt the same time oscillations of large neuronal populations have been observed with local field potentials (LFP) or EEG measurements 9, 10, 11 and these oscillations are classified into discrete frequency bands spanning single to tens of Hertz. In all cases, however, voltage gated sodium channels played a central role, as application of tetrodotoxin (TTX) abolished the sub-threshold oscillations 6, 8. Interestingly, these mechanisms are activated differentially when a cell is hyperpolarized (I h is activated) or depolarized (I M is activated). In the case of hippocampal pyramidal cells, the underlying ionic mechanisms are related to a slow hyperpolarization-activated cation current (often referred to as I h current), a slow activating potassium current (so called M-current as it acts through muscarinic receptors) and an instantaneously activating, inwardly rectifying potassium current. This phenomenon was reported in various cell populations such as hippocampal pyramidal neurons 6, 7 and pyramidal cells of the amygdala olfactory cortex 8. The natural frequency has been shown to be voltage dependent and it can shift at both depolarized and hyperpolarized membrane potentials 6, 7, 8. The ability to resonate at particular frequencies has been observed in many experimental preparations 4, 5. Single neurons integrate input to generate action potentials, however they can also display damped sub-threshold oscillations giving them complex dynamics and sensitivity to temporal patterns of input. However, it is unclear how these regions of enhanced connectivity or biased input can lead to virtually instantaneous, dynamically evolving and robust separation of activity patterns, which in turn encode for functionally diverse information content, while embedded within the same interconnected group of neurons. Correlations within these activity patterns lead to network rewiring, where the connections between neurons encoding the same functional pattern are strengthened. In terms of neuronal processing, many proposed coding schemes are based on either the firing rate or the temporal structure of spikes in response to a stimulus 1, 2, 3. What aspects of this patterning represent the information content? How do these network wide representations robustly emerge out of the dynamical properties of individual neurons? How are distributed features dynamically bound together, while at the same time segmented out from the rest of the network, to form a coherent representation? These questions not only pertain to the brain, but also to the formation of distributed representations in any network. However, basic questions remain unanswered. The brain forms a complex interconnected network in which spatio-temporal patterns of neuronal activity are thought to underlie its information processing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
